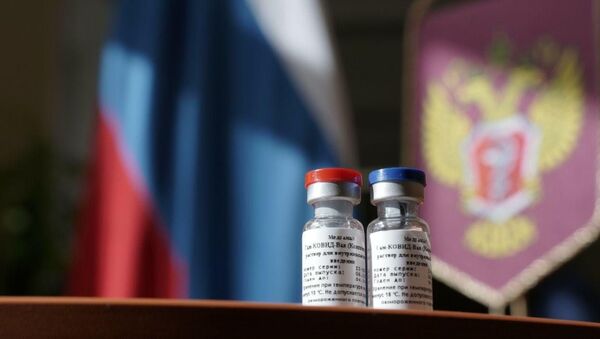
Earlier in the day, the Russian Health Ministry registered the first coronavirus vaccine in the world. It was developed jointly by the Gamaleya Research Institute and the Russian Defense Ministry.
“The WHO is aware that a COVID-19 vaccine has been registered in the Russian Federation's national medicines registry … WHO is in touch with Russian scientists and authorities, and looks forward to reviewing details of the trials,” the organization said.
The leading health organization welcomed all advances in the COVID-19 vaccine research and noted that acceleration of the development should be done in accordance with established processes to ensure safety.
“The WHO welcomes all advances in COVID-19 vaccine research and development. At the global level, the WHO has been involved in guiding and accelerating R&D efforts since January 2020.
Accelerating vaccine research should be done following established processes through every step of development, to ensure that any vaccines that eventually go into production are both safe and effective. Any safe and effective pandemic vaccine will be a global public good, and the WHO urges rapid, fair and equitable access to any such vaccines worldwide,” the organization underlined.
Last week, the WHO told Sputnik that a vaccine against coronavirus should go through the prequalification process, which involves “a rigorous review and assessment of all required safety and efficacy data gathered through clinical trials.”