https://sputnikglobe.com/20210204/uk-launches-trial-giving-volunteers-alternating-pfizer-and-astrazeneca-covid-19-vaccine-doses-1081976694.html
UK Launches Trial Giving Volunteers Alternating Pfizer and AstraZeneca COVID-19 Vaccine Doses
UK Launches Trial Giving Volunteers Alternating Pfizer and AstraZeneca COVID-19 Vaccine Doses
Sputnik International
MOSCOW (Sputnik) - The United Kingdom has launched recruitment for the world's first clinical trial that will see volunteers given alternating doses of... 04.02.2021, Sputnik International
2021-02-04T06:58+0000
2021-02-04T06:58+0000
2023-05-28T15:11+0000
https://cdn1.img.sputnikglobe.com/img/07e4/0c/08/1081393011_0:51:3040:1761_1920x0_80_0_0_7bd315390b670b05a99f04142687d1c4.jpg
united kingdom (uk)
Sputnik International
feedback@sputniknews.com
+74956456601
MIA „Rosiya Segodnya“
2021
Sputnik International
feedback@sputniknews.com
+74956456601
MIA „Rosiya Segodnya“
News
en_EN
Sputnik International
feedback@sputniknews.com
+74956456601
MIA „Rosiya Segodnya“
Sputnik International
feedback@sputniknews.com
+74956456601
MIA „Rosiya Segodnya“
newsfeed, coronavirus, united kingdom (uk), pfizer, pfizer, astrazeneca
newsfeed, coronavirus, united kingdom (uk), pfizer, pfizer, astrazeneca
UK Launches Trial Giving Volunteers Alternating Pfizer and AstraZeneca COVID-19 Vaccine Doses
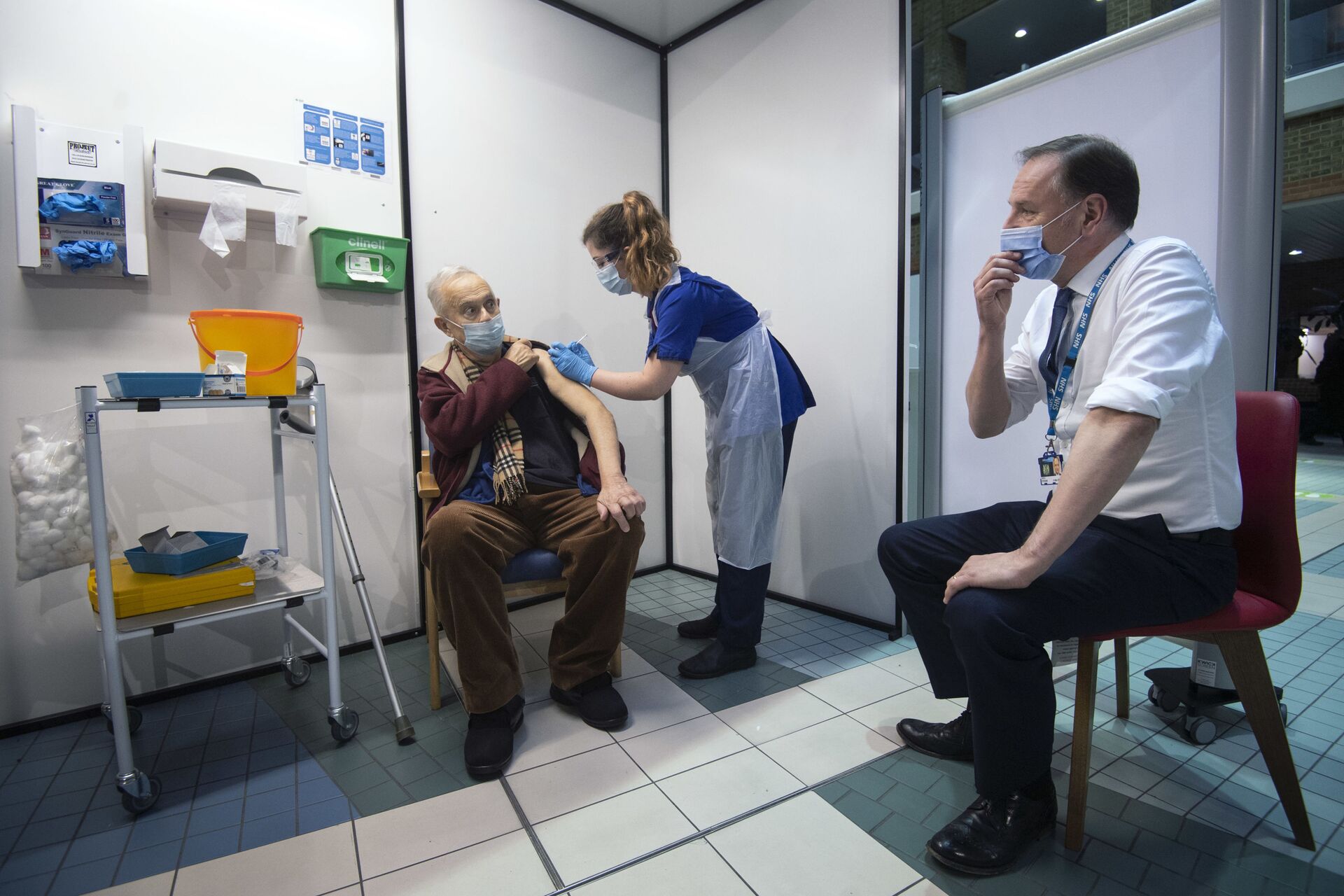
06:58 GMT 04.02.2021 (Updated: 15:11 GMT 28.05.2023) MOSCOW (Sputnik) - The United Kingdom has launched recruitment for the world's first clinical trial that will see volunteers given alternating doses of COVID-19 vaccines produced by AstraZeneca/Oxford University and Pfizer/BioNTech, the Department of Health and Social Care said Thursday.
"Backed by £7 million [$9.5 million] of government funding, the study will be the first in the world to determine the effects of using different vaccines for the first and second dose – for example, using Oxford University/AstraZeneca’s vaccine for the first dose, followed by Pfizer/BioNTech’s vaccine for the second", the department said in a press release.
The study is being run by the National Immunisation Schedule Evaluation Consortium and will also assess the effects of giving participants a mixed-vaccine regimen at varying intervals.
"This is a hugely important clinical trial that will provide us with more vital evidence on the safety of these vaccines when used in different ways", Nadhim Zahawi, the UK's minister for COVID-19 vaccine deployment, said in the press release.
The government said that there were no current plans to alter the UK's national COVID-19 vaccination programme, which sees individuals receive two shots of the same vaccine.
Russian vaccine manufacturers have also raised the possibility of conducting mixed-dose trials. Kirill Dmitriev, the CEO of the Russian Direct Investment Fund, said in early January that plans were in place to conduct mixed Sputnik V-AstraZeneca dosing regimen trials.